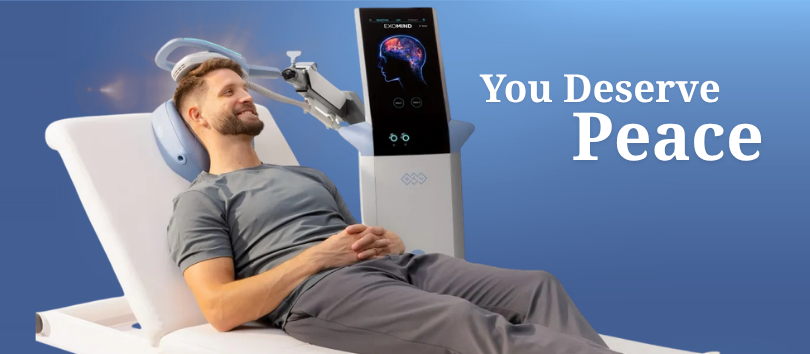
FDA-Cleared Indication: EXOMIND® is intended for the treatment of Major Depressive Disorder (MDD) in adult patients who have failed to achieve satisfactory improvement from prior antidepressant medication in the current episode. EXOMIND® should only be used under the continued supervision of a physician or licensed practitioner.
Additional Conditions: Certain conditions discussed on this page, including anxiety, PTSD, emotional eating, and cognitive fatigue, reflect clinician experience, published research, and international regulatory approvals (Health Canada, CE Mark). These are not independently FDA-cleared indications for this device in the United States. Treatment for these conditions is provided at the discretion of your healthcare provider.
Contraindications: EXOMIND® is not appropriate for individuals with metallic or electronic implants in or near the head (including cochlear implants, deep brain stimulators, and cardiac pacemakers), a history of seizures or epilepsy, or those on certain anticoagulation therapies. A safety screening is required before treatment begins. Please consult your healthcare provider for a full list of contraindications, warnings, and precautions.
Results: Results based on published TMS clinical data. Patient results and experience may vary. EXOMIND® is not a guarantee of symptom improvement.The information on this website is for informational purposes only and does not constitute medical advice, diagnosis, or treatment. Always seek the advice of a qualified healthcare provider with any questions regarding a medical condition.
EXOMIND®, ExoTMS™, and BTL® are trademarks and/or registered trademarks of BTL Industries, Inc. The products, the methods of manufacture, or their use may be protected by one or more U.S. or foreign patents or pending applications.
See
btlnet.com/patents. © 2026 Health Restoration and HR Pain Management •
Terms & Conditions •
Privacy Policy 
.png)
.png)
.svg)
.png)
.svg)
.png)
.png)
.png)
.png)
.png)













.png)



